Precise monitoring of the lung
X-rays and fluorescence imaging of the lungs of a mouse enable tracking of drug delivery
2020-01-31 – News from the Physics Department
Drugs inhaled by patients are well established as effective treatments for lung diseases such as chronic obstructive pulmonary disease (COPD), asthma or infections. In the future, other drugs might be administered in this way – for example cancer medications in which the effective ingredient is contained in nanoparticles. “Nanoparticle-based drugs have many advantages,” says Otmar Schmid, who heads a working group at the Institute for Lung Biology and Disease at the Helmholtz Zentrum München. “For example, the effective ingredients cannot be broken down as easily by enzymes because they are embedded in the nanoparticle. The nanoparticles can also target the diseased cells more specifically by attaching ligands that dock onto them.” This would mean a more focused delivery of the effective ingredient to the diseased tissue, which would significantly reduce the side effects.
Researchers in Otmar Schmid’s working group have now teamed up with colleagues at the Physics Department and the Munich School of BioEngineering (MSB) at TUM to present a combination of imaging techniques that can show in detail how the delivery of a drug in the lung of a mouse depends on the way the drug is administered. The results of such studies are intended to provide important insights for the development of lung medications for humans.
Lung filmed at particle accelerator

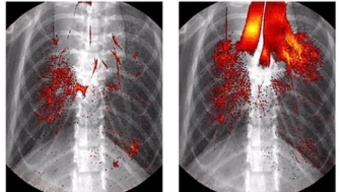
The TUM researchers contributed two techniques that use X-rays. With one of the techniques, they were able to record video images showing how drugs inhaled by a mouse spread from the air tube throughout the lung in real time. The other technique generated 3D images of the entire lung. Instead of administering a real drug, they used a solution containing nanoparticles, but with no active ingredient, mixed with an iodinated medical contrast agent that is clearly visible in X-ray images.
The researchers produced the film using X-ray images of the mouse lung taken at the same point in the breathing cycle. “By using the propagation effect of the X-rays, we were able to create a clear visualization of the tissue structures in the lung,” says Regine Gradl, who performed the measurements as part of her doctoral research at the TUM Chair of Biomedical Physics. “In conventional X-ray images, we would not have seen much more than the ribcage and the iodinated contrast agent.” The propagation effect uses the fact that the X-rays are slightly deflected as they pass through the sample under investigation, which can cause interference between some of the rays behind it. These interference effects reveal additional details in the X-ray image, in particular at the borders of soft tissue, for example where air meets lung tissue.
Until recently, these investigations were possible only using synchrotron light sources, i.e. complex particle accelerators that are often several hundred meters in diameter. It was only these facilities that could produce X-rays with the necessary intensity and the required coherence to produce the interference effects. With the Munich Compact Light Source (MuCLS), the world’s first mini-synchotron, which went into operation a few years ago, it is now possible to produce X-rays with these characteristics in a normal-sized laboratory. Installed in the main MSB building in Garching, it has a 3 by 5 meters footprint and is just 2 meters tall. It gives researchers flexibility in scheduling extensive investigations using intensive, coherent X-rays. “Synchrotron light sources are in such heavy demand among researchers that one has to plan their experiments far in advance and complete them within just a few days,” says Gradl. With conventional laboratory X-ray equipment, however, measurements of this type would not have been possible.
Fluorescence shows precise distribution


The scientists at the Helmholtz Zentrum München were subsequently able to apply fluorescence techniques to the explanted lung to show how the inhaled particles were distributed to the cells. For this investigation, fluorescent nanoparticles were used. When stimulated with light, these particles glow to reveal where the solution is located in the lung. The researchers achieved highly precise results with light sheet fluorescence microscopy (LSFM). In this method, only a very thin slice of the sample is illuminated at any time. However, an exact 3D image of the entire lung is created, including the nanoparticles localized in it, by illuminating successive slices.
Unique combination
“With the combination of X-ray and fluorescence techniques, we were able to observe both the dynamic delivery process of lung medication as well as the final distribution for the first time,” says Schmid. In the future this should make it possible, for example, to decide exactly how nanoparticle-based drugs will reach the target tissues. But even these initial experiments have provided important insights. The researchers compared two methods for delivering drugs to the lung in pre-clinical trials: the inhalation of droplets and the instillation of bulk liquids, in which the solution is injected into the lung through the trachea with a tube. They were able to show how it is possible for the instilled liquid to be distributed down to the finest bronchioles in the lung. “It initially pools in the upper breathing passages until it blocks them. With the next breath it is then distributed as a spray throughout the lung,” says Schmid. “In contrast to drugs administered by inhalation, however, it does not reach the lowest regions of the lung.”