DNA origami surpasses important thresholds
Building virus-sized structures and saving costs through mass production
2017-12-07 – News from the Physics Department

Viruses encapsulate their genetic material in a shell comprising a series of identical protein building blocks. The hepatitis B virus capsule, for example, comprises 180 identical subunits, a typical case of “prefabricated” construction deployed frequently in nature.
The team led by Hendrik Dietz, Professor of Biomolecular Nanotechnology at the TU Munich has now transferred viral construction principles to DNA origami technology. This allows them to design and build structures on the scale of viruses and cell organelles.
The technology builds on a long single strand that is appended to a double-stranded structure using short staple sequences. “The double-stranded structure is energetically sufficiently stable so that we can force the single strand into almost any shape using appropriately chosen counterparts,” explains Hendrik Dietz. “This way we can precisely design objects in the computer that are merely a few nanometers in size.”
Gears for nanomotors
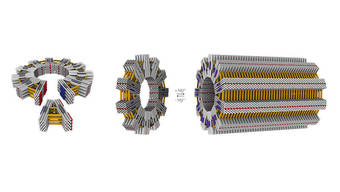
The Dietz lab commands techniques that allow them to further modify and insert chemical functionalities into objects by adding side groups. But, until now, the size of the objects remained in the nanometer realm. In the renowned scientific journal Nature, the team now describes how larger structures can be built using prefabricated parts.
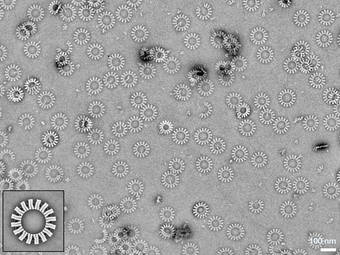
To this end, they first created V-shaped nano-objects. These have shape-complementary binding sites on their sides, allowing them to autonomously attach to each other while floating in a solution. Depending on the opening angle, they form “gears” with controlled number of spokes.
“We were thrilled to observe that, almost without exception, rings formed as defined by the opening angle,” says Hendrik Dietz. “Decisive for the ability to build objects of this size and complexity is the precision and rigidity of the individual building blocks. We had to reinforce individual elements with crossbars, for instance.”
Construction of microtubes
To further exploit the construction principle, the team created new building blocks that had “glue joints” not only on the sides, but also slightly weaker ones on the top and bottom. This allows the “nano-gears” to form long tubes using the additional docking sites in a second step.
“At lengths of one micrometer and a diameter of several hundred nanometers, these tubes have reached the size of some bacteria,” explains Hendrik Dietz. “And we can use the architecture of individual elements to determine features of the overall structure.”
Building polyhedral structures
Inspired by the symmetries and the hierarchical design of viruses, the researchers also attempted to build closed cage structures. “A potential future application of artificial cages is the transport of medication in the body,” explains Hendrik Dietz. “Here, the goal is to release active agents only at specific desired locations, sparing the rest of the body.”
Using the principles already applied to the structures described before, the team now constructed new elements they hoped would assemble in a self-limiting fashion into cage structures under the right conditions. According to these strategies a triangular middle section and three V-shaped elements give rise to a three-pronged building element.
Depending on the opening angle of the V, a defined number of these units merge to form tetrahedral, hexahedral or dodecahedral structures in a second step. The final structures integrate up to 1.8 million addressable DNA base pairs at user-defined positions. For the first time, these discrete-size cages attain molecular weights and sizes comparable to that of viruses and small cell organelles.
Cost-effective mass production

To date, manufacturing processes have limited the scope of application to those requiring only small amounts of material. The fact that only a few micrograms can be manufactured with conventional methods precludes many potential medical and materials science applications.
The bottleneck is the short staple strands that must be chemically produced base by base. The main strand obtained from bacteriophages, on the other hand, can be produced on a large scale using biotechnological processes.
That is why the team led by Hendrik Dietz refined so-called DNA enzymes, a discovery stemming from synthetic biotechnology. These are DNA strands that break apart at specific positions when exposed to a high concentration of zinc ions.
They joined the short staple sequences to a long strand using two modified DNA enzymes each. “Once precisely assembled with a specific base sequence, these combined strands can be reproduced in a biotechnological process, as with single strands of bacteriophage DNA,” says Dietz, explaining the key feature of the process.
Biotechnological production on a large scale
Both the main strand and the secondary strand, comprising DNA enzymes and the staple sequences, were successfully produced using a high cell density process with bacteria. The process is scalable and thus amenable to high volume production of the main strands and staples. Increasing the zinc ion concentration after DNA isolation releases the short staple sequences, which then fold the main strand into the desired shape.
Extensive investigations of the reaction mechanisms in collaboration with colleagues at the Institute of Biochemical Engineering showed that this is possible even on a large scale. At the TUM Research Center for Industrial Biotechnology in Garching, scientists have now produced multiple grams of four different DNA origami objects. Scaling up the process to a cubic meter scale is now within grasp.
“The interplay of biotechnology and process technology has thus enabled setting a truly fundamental milestone on the path to future applications in DNA nanotechnology,” says Professor Dirk Weuster-Botz, Chair of the Institute of Biochemical Engineering.
The research was funded by the European Research Council, the German Research Foundation (DFG) through the Gottfried-Wilhelm-Leibniz Program, the collaborative research project SFB 863, the Excellence Cluster for Integrated Protein Science Munich (CIPSM) and Nanosystems Initiative Munich (NIM), the International Graduate School of Science and Engineering, the Institute for Advanced Study of the Technical University of Munich using funds from the DFG and the European Community, the BioOrigami project of the German Federal Ministry of Education and Research (BMBF), and the Bosch Research Foundation.
Publications
- Gigadalton-scale shape-programmable DNA assembliesKlaus F. Wagenbauer, Christian Sigl und Hendrik DietzDOI: 10.1038/nature24651
- Biotechnological mass production of DNA origamiFlorian Praetorius, Benjamin Kick, Karl L. Behler, Maximilian N. Honemann, Dirk Weuster-Botz und Hendrik DietzDOI: 10.1038/nature24650