Turbulenzen im Kristall
Nachrichten aus dem Physik-Department - 2013-03-05
Ultrakurzzeitphysiker beobachten, welche Auswirkungen Elektronenverschiebungen im Kristall auf den gesamten Festkörper haben.

Wenn auf einen Festkörper intensives Licht trifft, dann kommt sein atomares Innenleben in Bewegung. In Titanoxid, einem Halbleiter, beobachtete nun ein Team von Ultrakurzzeitphysikern vom Max-Planck-Institut für Quantenoptik in Garching (MPQ), der Technischen Universität München (TUM), dem Fritz-Haber-Institut in Berlin (FHI) und der Universität Kassel wie sich die Anordnung von Elektronen und Atomkernen verändert, wenn starkes Laserlicht auf den Titandioxid-Kristall trifft. Die Forscher wiesen nach, dass selbst kleine Veränderungen in der Elektronenverteilung, ausgelöst durch eine Anregung mit ultrakurzen Laserpulsen, eine große Wirkung auf das gesamte Kristallgitter haben können.
Das Wissen um die Wechselwirkung zwischen Licht und Materie in atomaren Dimensionen gleicht einer Landkarte mit vielen weißen Flecken. Unzählige Phänomene harren hier ihrer Entdeckung. Einen neuen, bisher unbekannten Aspekt des Licht-Materie-Wechselspiels in Kristallen hat ein Team aus Ultrakurzzeitphysikern vom Max-Planck-Institut für Quantenoptik in Garching, der Technischen Universität München, dem Fritz-Haber-Institut in Berlin und der Universität Kassel mit Laserpulsen von wenigen Femtosekunden (eine Femtosekunde ist ein Millionstel einer milliardstel Sekunde) untersucht.
Die Physiker schickten einen intensiven, ultravioletten Laserpuls mit einer Dauer von weniger als fünf Femtosekunden auf den Titandioxid-Kristall (bestehend aus Titan und Sauerstoffatomen). Dadurch gerieten die Valenzelektronen der Atome in Bewegung und erhitzten sich auf mehrere Tausend Grad Celsius. Valenzelektronen sind locker an Atome gebundene Elektronen, sie treten in starke Wechselwirkung miteinander und bilden dadurch eine Art Klebstoff, der die Atomrümpfe in einem Kristallgitter zusammenhält. Eigenschaften eines Materials, wie etwa die elektrische Leitfähigkeit, die optischen Eigenschaften oder die Gitterstruktur, werden durch das permanente Wechselspiel zwischen der Position der Atomrümpfe und deren Valenzelektronen bestimmt.
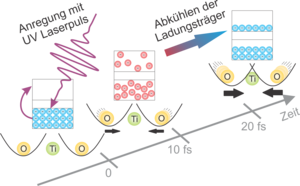
Wenige Femtosekunden nach dem ersten Laserpuls schickten die Physiker einen zweiten, etwas schwächeren Puls auf den Kristall. Dieser wurde an der Oberfläche reflektiert und gab den Forschern dadurch Auskunft über die Veränderungen, die der erste Puls im Kristall hervorgerufen hatte: Das starke Licht des ersten Pulses erhitzte nicht nur die Valenzelektronen, es veränderte auch deren Position im Atomgitter. Die Elektronendichte wurde in der Umgebung der Sauerstoffkerne verringert und in der Umgebung der Titankerne erhöht. Die Verschiebung des Gleichgewichts bedeutete wiederum, dass sich die Ruheposition der Sauerstoffatome relativ zur Ruheposition der Titanatome verschob. Letztendlich begannen die Sauerstoff-Atomrümpfe zu schwingen. Dieser Effekt lässt sich mit einer Kugel (Sauerstoffatom) in einer Schale (gesamter Kristall) veranschaulichen. Im Grundzustand befindet sich die Kugel in der Mitte am tiefsten Punkt der Schale. Die Anregung der Elektronen bewirkt eine schlagartige Verschiebung der Schale, die Kugel beginnt um die neue Gleichgewichtslage zu oszillieren.
Bei den Experimenten beobachteten die Physiker einen überraschenden Effekt: Nach der Lichtanregung kühlten die Elektronen innerhalb von rund 20 Femtosekunden auf Raumtemperatur ab. Der Kristall wurde während dieser kurzen Zeit nur minimal erwärmt. Die räumliche Verteilung der Valenzelektronen jedoch veränderte sich markant. Als Konsequenz daraus verschob sich auch die Ruheposition der Atome im Kristallgitter noch um ein ganzes Stück weiter.Eine solche Abhängigkeit der Kristallstruktur von der Temperatur angeregter Elektronen war schon lange theoretisch vorhergesagt. Nun gelang der experimentelle Nachweis. Das Ergebnis zeigt, dass der Gleichgewichtszustand des Festkörpers auch auf kleine Änderungen der Elektronenverteilung extrem stark reagiert. Dieses Wissen könnte später beim Design neuer Materialien von großem Nutzen sein.
Text: Thorsten Naeser
Publikation
Ultrafast evolution of the excited-state potential energy surface of TiO2 single crystals induced by carrier cooling
Elisabeth M. Bothschafter, Alexander Paarmann, Eeuwe S. Zijlstra, Nicholas Karpowicz, Martin E. Garcia, Reinhard Kienberger und Ralph Ernstorfer
Kontakt
Elisabeth Bothschafter und Prof. Dr. Reinhard Kienberger
Technische Universität München
Physik-Department E11
85747 Garching, Germany
Tel.: +49 89 289-12840
Fax: +49 89 289-12839
E-Mail: elisabeth.bothschafter@tum.de, reinhard.kienberger@tum.de